Background:
Exposure to ionizing radiation is associated with the development of therapy-related myeloid neoplasms (t-MNs). Several distinct clinical and cytogenetic subtypes of t-MN have been recognized, and are found to be closely associated with various cytotoxic/genotoxic therapies. However, characteristics of t-MNs that follow radiation therapy (RT) alone are not as clear. We examined clinical, epidemiologic, cytogenetic, and molecular characteristics of 108 cases of t-MN in patients (pts) who received RT alone for an antecedent disease.
Methods:
Retrospective chart review was done to identify pts in our institution's t-MN database that had received RT alone prior to development of t-MN. Clinical characteristics, indication for RT, latency period between radiation and development of t-MN (LP), cytogenetic features, somatic and germline mutation data, treatment, and overall survival (OS) after development of t-MN were collected. LP and OS were both analyzed using Kaplan-Meier analysis.
Results:
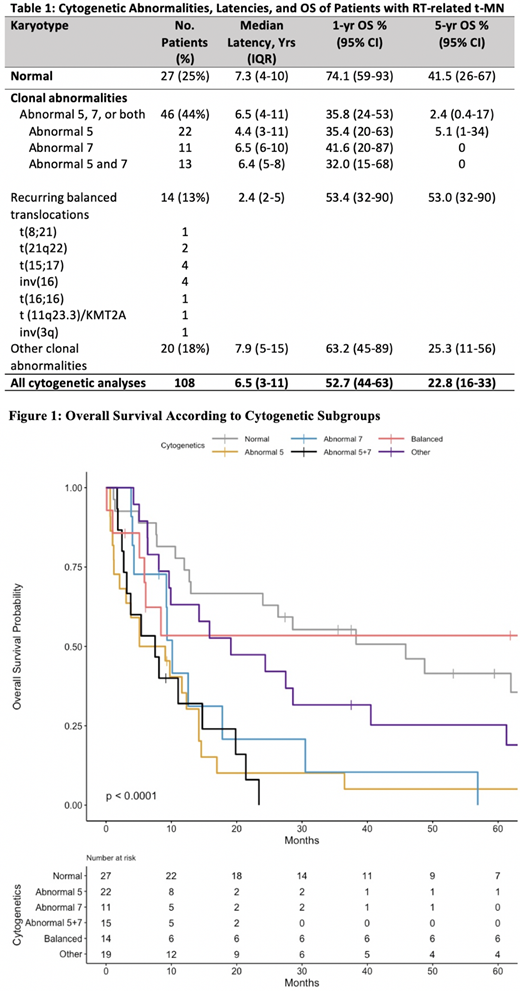
108 consecutive pts who developed an RT-related t-MN (48 with t-MDS at diagnosis, 60 with t-AML) were identified from 1971-2020. 99 pts had a primary solid tumor [most common: prostate/testicular (44), breast (30), and gynecological (15)], 4 had a primary lymphoid malignancy, and 5 received radiation for a non-malignant condition. 27 pts had a normal karyotype, 22 had an abnormality of chromosome 5, 11 had an abnormality of chromosome 7, 13 had abnormalities of both 5 and 7, 14 had recurring balanced translocations, and 20 had other clonal abnormalities (Table 1). Median LP for the entire cohort was 6.5 years. The shortest LP was in pts with recurring balanced translocations (median 2.4 years) (Table 1).Treatment course was available for 85 pts; 25 (29%) received intensive remission induction with cytarabine + anthracycline, 35 (44%) received hypomethylating agent-based therapy, 18 (21%) received only supportive care, and 5 (6%) received other therapies. 15 pts (18%) underwent allogeneic hematopoietic cell transplantation. Median OS was 12.7 months; 1-year OS was 53%, and 5-year OS was 23%. 1-year OS and 5-year OS were inferior in pts with an abnormality in chromosome 5 (p<0.01), abnormality in chromosome 7 (p=0.01), and abnormalities in both 5 and 7 (p<0.01) (Figure 1). Pts with chromosome 5 and/or 7 abnormalities had a median OS of 9.1 months.
Somatic next-generation sequencing (NGS) data were available for 15 pts at diagnosis of t-MN. The most common somatic mutations were ASXL1 (n=4), IDH2 (3),TET2 (3), U2AF1 (3), TP53 (2), NPM1 (2), FLT3- ITD (2), NRAS (2), STAG2 (2), SRSF2 (2), and CBL (2). Germline testing was available starting in 2011 and was performed in 16 pts. Pathogenic/likely pathogenic variants in CHEK2 p.Ile157Thr (no somatic mutations), DDX41 p.Pro258Leu (somatic ASXL1 mutation), or PALB2 p.Arg170Ilefs*14 (somatic NGS not done) were identified in 3 individuals (18%). All 3 pts had a normal karyotype.
Conclusions:
RT-related t-MNs have clinical/cytogenetic features, LPs, and outcomes similar to pts who have been exposed to alkylator-based therapy, with abnormalities in chromosome 5 and/or 7 (44%) being the most common subtype, and a median LP of 4.4-6.5 years. OS is poor, despite intensive therapies. Pts with normal karyotype or balanced rearrangements have better OS. Pts with RT-related APL, CBF AML, or normal-karyotype, NPM1-mutated AML have outcomes similar to pts with de novo disease. In addition, 18% of pts who underwent germline testing had a pathogenic variant. Better characterization of pathogenic somatic mutations in these patients and identification of germline predisposition mutations may provide new insights into the etiology of RT-related t-MNs and lead to more successful therapies.
Godley:UptoDate, Inc.: Honoraria; Invitae, Inc.: Membership on an entity's Board of Directors or advisory committees. Le Beau:American Cancer Society: Membership on an entity's Board of Directors or advisory committees; Varian Medical Systems: Membership on an entity's Board of Directors or advisory committees. Larson:Novartis, Takeda, CVS/Caremark, Celgene, Amgen, Epizyme: Consultancy; Astellas, Celgene, Daiichi Sankyo, Novartis, Rafael Pharmaceuticals, Cellectis, Forty Seven: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal